Robert Boyle (1627-1691)
Boyle was born in Lismore, County Waterford, Ireland, and he was greatly known as a natural philosopher, chemist, physicist, and inventor. He is pinned as one of the pioneers of modern experimental scientific method and founders of chemistry, thanks to his work in his book "The Sceptical Chymist" in 1661. Here he spoke of his hypothesis that said matter contained atoms and clusters of atoms in motion, and that every phenomenon was the result of collisions of particles in motion. Boyle was also the founder of "The Royal Society of London for the Improvement of Natural Knowledge" in 1660. Numbers of people with interests in science went to the meeting held in Gresham College. This was the only way Boyle could interact with the world of science, later he went to Oxford University and worked at establishing chemistry as a mathematical science based on the mechanistic theory of matter. He discovered the relationship between the absolute pressure and volume of a gas, if the temperature is kept constant, or better known as the Boyle's Law (1661-1662), sometimes known as the Marriotte's Law. He figured this after 3 years of experimenting with an air pump and Robert Hooke as his assistant.
Real life example
A simple example of a real life situation would be the popping of a balloon! When we squeeze the balloon (putting pressure on it), the volume inside is actually reducing. Since the balloon can't handle the extra pressure, it bursts.
Another example would be the bubbles a scuba diver exhales as they approach the surface of the ocean, the pressure put forth by the weight of the water decreases with depth. Therefore the volume of the bubbles increases as they rise.
Another example would be the bubbles a scuba diver exhales as they approach the surface of the ocean, the pressure put forth by the weight of the water decreases with depth. Therefore the volume of the bubbles increases as they rise.
Algebraically from the ideal gas law
Boyle's law can be driven from the Ideal Gas law which is PV=nRT simply by taking off the right side of the equation!
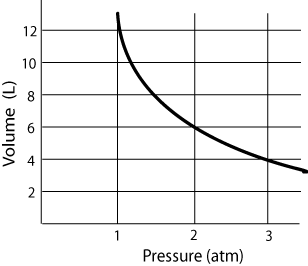
Graph
- P is inversely related to V
- Pressure (atm) and Volume (Liters)
- As volume decreases, pressure increases, and vice versa
Remember the formula is P1v1=p2v2 (T is constant)
Let's practice with an example problem!
Here we can just plug the information into the formula!
(1.00 atm)(1.56L) = (3.00 atm) (x)
Solve algebraically!
1.56 = (3.00 atm) (x)
x = .52
Now try some on your own!
- A gas occupies 1.56 L at 1.00 atm. What will be the volume of this gas if the pressure becomes 3.00 atm?
Here we can just plug the information into the formula!
(1.00 atm)(1.56L) = (3.00 atm) (x)
Solve algebraically!
1.56 = (3.00 atm) (x)
x = .52
Now try some on your own!
practice problems
- A gas occupies 4.31 liters at a pressure of 0.755 atm. Determine the volume if the pressure is increased to 1.25 atm.
- 600.0 mL of a gas is at a pressure of 8.00 atm. What is the volume of the gas at 2.00 atm? (change mL to L by multiplying number by 1000)
- 4.00 L of a gas are under a pressure of 6.00 atm. What is the volume of the gas at 2.00 atm?
- A 40.0 L tank of ammonia has a pressure of 8.00 atm. Calculate the volume of the ammonia if its pressure is changed to 12.0 atm while its temperature remains constant.
- A container of oxygen has a volume of 30.0 mL and a pressure of 4.00 atm. If the pressure of the oxygen gas is reduced to 2.00 atm and the temperature is kept constant, what is the new volume of the oxygen gas? (Remember to convert mL to L)
Answer Key
- (0.755 atm) (4.31 liters) = (1.25 atm) (x); x = 2.60 L
- (8.00 atm) (600.0 mL) = (2.00 atm) (x); x = 2.40 L
- (6.00 atm) (4.00 L) = (2.00 atm) (x); x = 12.00 L
- (8.00 atm) (40.0 L ) = (12.0 atm) (x); x = 26.67 L
- (4.00 atm) (30.0 mL) = (2.00 atm) (x); x = .06 L
Cited sources
"ChemTeam: Gas Law Problems- Boyle's Law." ChemTeam: Gas Law Problems- Boyle's Law. N.p., n.d. Web. 04 May 2014. <http://www.chemteam.info/GasLaw/WS-Boyle.html>.
"Robert Boyle." Wikipedia. Wikimedia Foundation, 05 Apr. 2014. Web. 04 May 2014. <http://en.wikipedia.org/wiki/Robert_Boyle>.
"Robert Boyle (1627-1691)." Campus Udayton. N.p., n.d. Web. 4 May 2014. <http%3A%2F%2Fcampus.udayton.edu%2F~hume%2FBoyle%2Fboyle.htm>.
By: Safa Kazi and Vera Jaeshcke
"Robert Boyle." Wikipedia. Wikimedia Foundation, 05 Apr. 2014. Web. 04 May 2014. <http://en.wikipedia.org/wiki/Robert_Boyle>.
"Robert Boyle (1627-1691)." Campus Udayton. N.p., n.d. Web. 4 May 2014. <http%3A%2F%2Fcampus.udayton.edu%2F~hume%2FBoyle%2Fboyle.htm>.
By: Safa Kazi and Vera Jaeshcke