Avagadro's Number
Avagadro's Number = 6.022 x 10^23
- It's the number of atoms or molecules in one mole
Here is a graphic organizer!
This chart can tell you how to get from one mole to another and convert them to liters, grams, and molecules!
- Liters to moles = divided by 22.4 tsp (multiply to go from moles to grams)
- To get to moles from grams, divide the grams by the molar mass (do opposite for vice versa)
- From atoms to moles, divide by Avagadro's number and vice versa
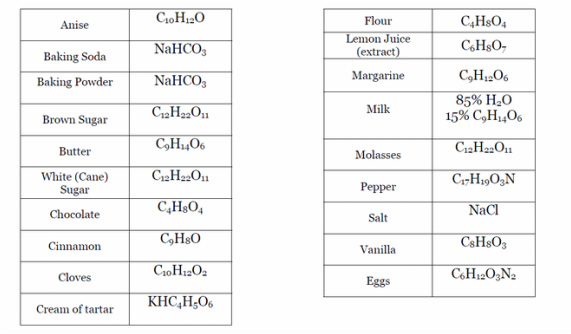
Here is an example of moles converting by using a recipe!
Using the chart above, you can convert the recipe below from moles to the proper cooking measurements:
(To find the grams, just find the molar mass of the corresponding formula and multiply by the moles)
Chocolate Chip Cookies (Converted to grams)
319.48g Flour 106.11g Packed Brown Sugar
2.81g Baking Soda 98.07g Eggs
2.1g Salt 9.43g Vanilla
145.5g Butter 338.69g Chocolate Chips
150.61g Sugar
Chocolate Chip Cookies (Converted to grams)
319.48g Flour 106.11g Packed Brown Sugar
2.81g Baking Soda 98.07g Eggs
2.1g Salt 9.43g Vanilla
145.5g Butter 338.69g Chocolate Chips
150.61g Sugar
Now you can reduce recipes and adjust the measurements
Anzac Biscuits(In this recipe, 0.63 moles of water has been reduced to 0.45-0.45 moles. Divided by 0.63 moles, it becomes 0.714, so you multiply the moles of the ingredients to get the right amount of moles for the reduced amount)
2.66 Flour x.714= 1.89 moles
1.77 Rolled oats= 1.26 moles
4.58 Brown sugar= 3.27 moles
1.48 shredded coconut= 1.06 moles
0.95 Almonds= .68 moles
0.96 Cranberries= .69 moles
1.19 Butter= .85 moles
0.89 Honey= .64 moles
0.63 Water= .45 moles
Oatmeal Chocolate Chip Cookies(In this recipe, 0.72 moles of Rolled Oats has been reduced to 0.50 moles of Rolled Oats)
4.26 Flour x.694= 2.95 moles
0.72 Rolled Oats= .5 moles
3.78 Butter= 2.62 moles
2 Eggs= 1.3 moles
0.76 Walnuts= .527 moles
2.5 Sugar= 1.908 moles
2.75 Brown Sugar= 1.9 moles
3.8 Chocolate Chips= 2.63 moles
0.25 Baking Soda= .1735 moles
0.2 Vanilla= .1388 moles
Chocolate Chip Cookies
(In this recipe, 3.8 moles of Chocolate Chips has been reduced to 2.5 moles of Chocolate Chips)
4.8 Flour x.658= 3.5 moles
3.78 Butter= 2.48 moles
0.76 Eggs= .5 moles
0.76 Walnuts= .5 moles
3.35 Sugar= 2.2 moles
3.66 Brown Sugar= 2.4 moles
3.8 Chocolate Chips= 2.5 moles
0.2 Baking Soda= .13 moles
0.25 Vanilla= .16 moles
1.06 Coco Powder= .697 moles
Drop Sugar Cookies with Walnuts:
(In this recipe, 5.33moles of flour has been reduced to 4.33 moles of flour)
5.33 Flour x.812= 4.3 moles
2.35 Butter= 1.9 moles
0.48 Egg= .38 moles
0.96 Walnuts= .77 moles
4.19 Sugar= 3.4 moles
0.25 Baking Soda= .20 moles
0.31 Vanilla= .812 moles
2.66 Flour x.714= 1.89 moles
1.77 Rolled oats= 1.26 moles
4.58 Brown sugar= 3.27 moles
1.48 shredded coconut= 1.06 moles
0.95 Almonds= .68 moles
0.96 Cranberries= .69 moles
1.19 Butter= .85 moles
0.89 Honey= .64 moles
0.63 Water= .45 moles
Oatmeal Chocolate Chip Cookies(In this recipe, 0.72 moles of Rolled Oats has been reduced to 0.50 moles of Rolled Oats)
4.26 Flour x.694= 2.95 moles
0.72 Rolled Oats= .5 moles
3.78 Butter= 2.62 moles
2 Eggs= 1.3 moles
0.76 Walnuts= .527 moles
2.5 Sugar= 1.908 moles
2.75 Brown Sugar= 1.9 moles
3.8 Chocolate Chips= 2.63 moles
0.25 Baking Soda= .1735 moles
0.2 Vanilla= .1388 moles
Chocolate Chip Cookies
(In this recipe, 3.8 moles of Chocolate Chips has been reduced to 2.5 moles of Chocolate Chips)
4.8 Flour x.658= 3.5 moles
3.78 Butter= 2.48 moles
0.76 Eggs= .5 moles
0.76 Walnuts= .5 moles
3.35 Sugar= 2.2 moles
3.66 Brown Sugar= 2.4 moles
3.8 Chocolate Chips= 2.5 moles
0.2 Baking Soda= .13 moles
0.25 Vanilla= .16 moles
1.06 Coco Powder= .697 moles
Drop Sugar Cookies with Walnuts:
(In this recipe, 5.33moles of flour has been reduced to 4.33 moles of flour)
5.33 Flour x.812= 4.3 moles
2.35 Butter= 1.9 moles
0.48 Egg= .38 moles
0.96 Walnuts= .77 moles
4.19 Sugar= 3.4 moles
0.25 Baking Soda= .20 moles
0.31 Vanilla= .812 moles
Stoichiometry probelms
Here are some practice stoichiometry problems!
1. You want to make 6 grilled cheese sandwiches (use the equation below, Bd = bread and Ch = Cheese) and you have 13 slices of bread and 5 slices of cheese. How many grilled cheese sandwich can you make? And what is the limiting reagent?
2Bd+Ch=Bd2Ch
For every one sandwich, you need two slices of bread and one slice of cheese (2 slices of bread + 1 slice of cheese = 1 sandwich)
13 slices of bread/ 2 = 5 sandwiches with cheese
Limiting reagent: Cheese (reason being because there isn't enough cheese to match the amount of bread)
2. You are having some friends over for dinner you want to make 8 cheese burgers, you have 8 hamburgers patties, 12 buns, 7 slices of cheese, 18 slices of tomato, 16 pieces of lettuce and 9 slices of onions. Use the equation below ( where buns = B, hamburger patty = H, Cheese = Ch, tomatoes = T, lettuce = L and onions = O) to determine how many cheese burgers you can make ( what's your theoretical yield?) and what the limiting reagent it?
B+H+Ch+2T+2L+O = BHChT2L2O
3. Balance the chemical equation below. Find the limiting reagent and the theoretical yield if there it one gram of each reagent.
LiOH + Co2 → LiCO3 + H2O
Balance: 2 LiOH + 3 Co2 → 2 LiCO3 + H2O
Molar Mass of LiOH: 23.948
Mole of LiOH: 1g/23.948 = 0.0417 moles
Output of Water: Ratio 1/2
Theoretical: 0.0208
Molar Mass of Co2: 44.01
Mole: 1g/ 44.01= 0.022 moles
Ratio: 3/2 = 0.03405
Safa and Vera
1. You want to make 6 grilled cheese sandwiches (use the equation below, Bd = bread and Ch = Cheese) and you have 13 slices of bread and 5 slices of cheese. How many grilled cheese sandwich can you make? And what is the limiting reagent?
2Bd+Ch=Bd2Ch
For every one sandwich, you need two slices of bread and one slice of cheese (2 slices of bread + 1 slice of cheese = 1 sandwich)
13 slices of bread/ 2 = 5 sandwiches with cheese
Limiting reagent: Cheese (reason being because there isn't enough cheese to match the amount of bread)
2. You are having some friends over for dinner you want to make 8 cheese burgers, you have 8 hamburgers patties, 12 buns, 7 slices of cheese, 18 slices of tomato, 16 pieces of lettuce and 9 slices of onions. Use the equation below ( where buns = B, hamburger patty = H, Cheese = Ch, tomatoes = T, lettuce = L and onions = O) to determine how many cheese burgers you can make ( what's your theoretical yield?) and what the limiting reagent it?
B+H+Ch+2T+2L+O = BHChT2L2O
3. Balance the chemical equation below. Find the limiting reagent and the theoretical yield if there it one gram of each reagent.
LiOH + Co2 → LiCO3 + H2O
Balance: 2 LiOH + 3 Co2 → 2 LiCO3 + H2O
Molar Mass of LiOH: 23.948
Mole of LiOH: 1g/23.948 = 0.0417 moles
Output of Water: Ratio 1/2
Theoretical: 0.0208
Molar Mass of Co2: 44.01
Mole: 1g/ 44.01= 0.022 moles
Ratio: 3/2 = 0.03405
Safa and Vera